DFT functional comparison¶
Every density-functional approximation is a model for the exchange-correlation energy \(E_{\text{xc}}[\rho]\). They climb a well-known hierarchy — “Jacob’s ladder” — trading cost for physics:
Rung |
Input |
Cost |
Examples |
|---|---|---|---|
1. LDA |
density \(\rho\) only |
cheapest |
Slater, SVWN, PW92 |
2. GGA |
\(\rho\) + \(\nabla\rho\) |
+~10% |
PBE, BLYP, PW91 |
3. meta-GGA |
+ $\tau = \sum_i |
\nabla\phi_i |
^2 / 2$ |
4. hybrid |
+ fraction of HF exchange |
+~2× |
B3LYP, PBE0, M06-2X |
5. range-separated hybrid |
+ long-range HF exchange |
+~3× |
ωB97X, CAM-B3LYP |
Going up the ladder usually improves accuracy at the cost of more compute per SCF step. vibe-qc currently supports rungs 1, 2, and 4 (LDA, GGA, hybrid) via named functionals; anything libxc catalogues is also reachable through its numeric ID. This tutorial sweeps a set and compares them on water.
A single-line functional swap¶
from vibeqc import Atom, Molecule, run_job
mol = Molecule([
Atom(8, [0.0, 0.00, 0.00]),
Atom(1, [0.0, 1.43, -0.98]),
Atom(1, [0.0, -1.43, -0.98]),
])
for label, functional in (
("HF", None),
("LDA (SVWN)", "SVWN"),
("GGA (PBE)", "PBE"),
("GGA (BLYP)", "BLYP"),
("hybrid B3LYP", "B3LYP"),
("hybrid PBE0", "406"), # via libxc ID
):
run_job(
mol,
basis="6-31g*",
method="rhf" if functional is None else "rks",
functional=functional,
output=f"water_{label.replace(' ', '_').lower()}",
write_molden_file=False,
)
Each call writes an .out file you can eyeball; below we’ll pull
the same numbers more compactly.
What the numbers say¶
Running the sweep above and extracting total energy, dipole, and frontier-orbital levels (HF/6-31G* experimental-geometry water):
| Method | \(E\) (Ha) | \(|\mu|\) (D) | \(\varepsilon_{HOMO}\) (eV) | \(\varepsilon_{LUMO}\) (eV) | gap (eV) | |—|—:|—:|—:|—:|—:| | HF | −76.00668 | 2.061 | −13.56 | +6.11 | 19.67 | | LDA (SVWN) | −75.83424 | 1.988 | −6.32 | +1.61 | 7.93 | | GGA (PBE) | −76.31284 | 1.914 | −6.23 | +1.65 | 7.88 | | GGA (BLYP) | −76.37826 | 1.892 | −6.11 | +1.50 | 7.60 | | hybrid (B3LYP) | −76.40105 | 1.946 | −7.95 | +2.19 | 10.14 | | hybrid (PBE0) | −76.31857 | 1.971 | −8.32 | +2.70 | 11.02 |
Experimental reference: dipole 1.855 D (gas phase); vertical ionisation potential 12.6 eV ≈ −\(\varepsilon_{HOMO}\) by Koopmans’ theorem.
Reading across:
Total energies drift — LDA is too high (electronic correlation is underestimated); GGAs drop ~0.3–0.5 Ha; B3LYP is the lowest. Absolute totals aren’t meaningful to compare across functionals — they live on different reference surfaces.
Dipole moments are well-behaved. HF overpolarises; GGAs pull back and undershoot slightly; hybrids land in between. All are within 0.2 D of the experimental value.
HF HOMO is unphysically deep: −13.56 eV vs. experimental IP of 12.6 eV. HF’s lack of correlation is compensated by Koopmans’ theorem only partially.
Pure DFT HOMOs are too shallow (−6 to −6.3 eV) — the classic DFT gap-underestimation problem driven by self-interaction error.
Hybrids sit in the middle because they mix in ~20–25% exact HF exchange, which partially corrects both the self-interaction and the overbound electrons. B3LYP (−7.95 eV) and PBE0 (−8.32 eV) are much closer to experiment.
B3LYP beats PBE on total energy because the LYP correlation functional is particularly well-parametrised for first-row chemistry. This is an empirical match, not a systematic improvement — don’t extrapolate to transition metals.
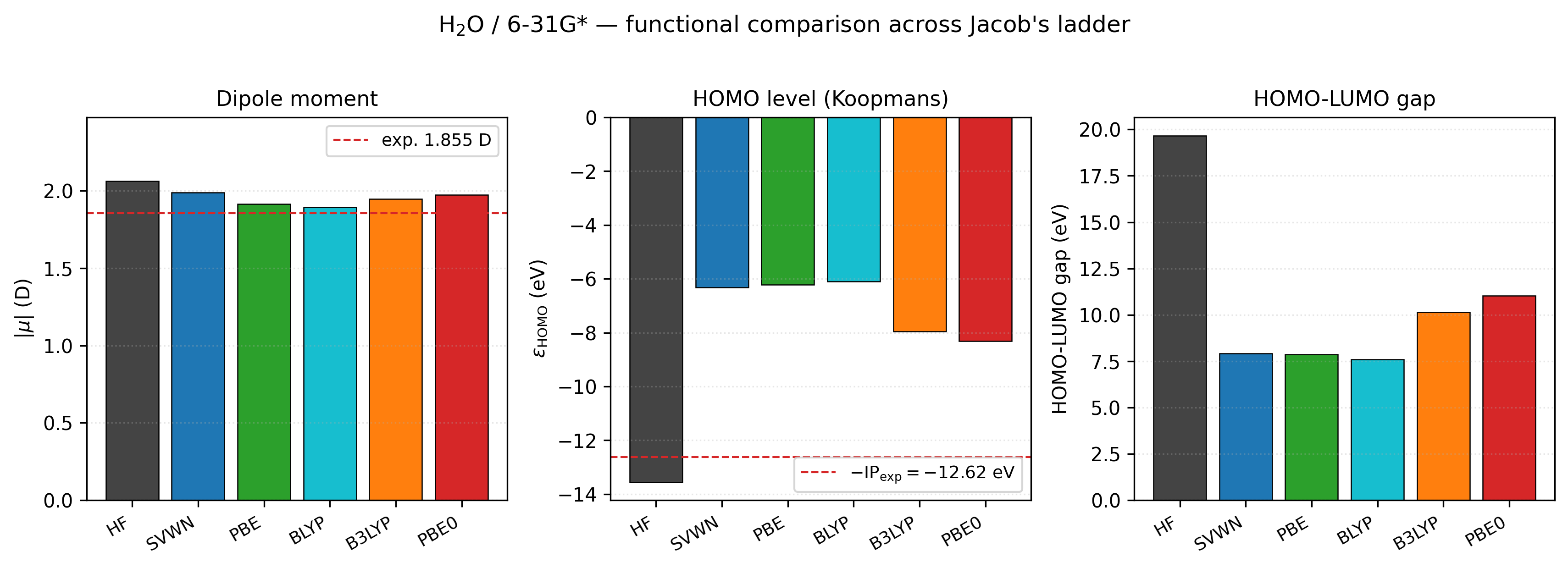
H₂O / 6-31G* across Jacob’s ladder, with experimental references
(red dashed). Left: dipole moment — every method is within 0.21 D
of the gas-phase value, with hybrids landing between HF
(over-polarised) and pure GGAs (slightly under-polarised). Centre:
HOMO level interpreted via Koopmans’ theorem against the
experimental vertical IP \(-12.6\) eV. HF is too deep by ~1 eV; pure
DFT is far too shallow (~6 eV); B3LYP and PBE0 — by mixing in
20–25% exact exchange — sit much closer to experiment. Right:
HOMO–LUMO gap. The well-known DFT gap-underestimation problem is
visible: pure GGAs give 7–8 eV, hybrids 10–11 eV, HF 19–20 eV;
none of these match the optical gap, which is what TDDFT exists to
compute. Regenerated by
examples/plots/water-functional-comparison.py.¶
Functional names vibe-qc knows¶
Via name (fast path):
Name |
Family |
HF exchange |
libxc components |
|---|---|---|---|
|
LDA |
0 |
Slater + VWN5 |
|
LDA |
0 |
Slater exchange only |
|
GGA |
0 |
X_PBE + C_PBE |
|
GGA |
0 |
X_B88 + C_LYP |
|
hybrid GGA |
0.20 |
XC_HYB_GGA_XC_B3LYP |
Via libxc ID (escape hatch for anything else libxc ships — 500+ functionals):
opts.functional = "406" # PBE0 (XC_HYB_GGA_XC_PBEH)
opts.functional = "402,101" # separate X and C IDs, comma-separated
Look up IDs at tddft.org/programs/libxc/functionals.
Currently out of reach in vibe-qc: meta-GGAs (SCAN, TPSS, M06-L) and range-separated hybrids (ωB97X, CAM-B3LYP). They need kinetic- energy-density evaluation and the range-separation machinery respectively — track status on the roadmap.
Cost across the ladder¶
On water / 6-31G* (11 basis functions; same machine, 12 OpenMP threads), SCF wall-clock time was roughly:
Method |
time (s) |
iters |
|---|---|---|
HF |
0.01 |
10 |
LDA (SVWN) |
0.23 |
9 |
GGA (PBE) |
0.33 |
9 |
GGA (BLYP) |
0.30 |
9 |
hybrid (B3LYP) |
0.34 |
9 |
hybrid (PBE0) |
0.32 |
9 |
Two observations:
HF is cheaper than DFT here because the DFT grid evaluation dominates at small N, where the four-index ERI sum is tiny. This flips at around 40–60 basis functions — pure DFT becomes cheaper than HF as the grid cost amortises.
Hybrids aren’t meaningfully more expensive at this size. The HF-exchange sum is a small fraction of the SCF cost when the system is this small. For bigger systems the hybrid overhead becomes real (~2× vs. pure DFT).
Picking a functional¶
A starter heuristic for general organic chemistry:
B3LYP — by far the most-published hybrid; “safe default” for geometries and binding energies on organic molecules. Known to underbind dispersion-dominated systems — always pair with DFT-D3.
PBE — the go-to for periodic calculations; faster than hybrids, reasonable for geometries. Use with D3(BJ) for molecular crystals.
PBE0 — hybrid cousin of PBE with 25% HF exchange. Better HOMO levels and reaction barriers than PBE; commonly used for TMs.
LDA — now mostly a teaching functional. Over-binds bonds, over-polarises, HOMO is too shallow. Still useful for quick screens on metallic systems where its simplicity is a virtue.
Rules of thumb that survive basis / method changes:
No functional is universally best. Benchmarks are the only defensible way to pick for a specific system class.
Most DFT errors cancel in relative energies. Reaction thermochemistry, conformational energies, and barriers are often within 1–2 kcal/mol across good GGA/hybrid choices. Absolute energies are much less stable.
Polar / ionic / H-bonded systems need dispersion. Add D3(BJ) — it’s a free lunch that always lowers the intermolecular error at negligible cost.
Transition metals are harder. B3LYP can be wrong by 10+ kcal/mol on spin states; PBE0, M06, ωB97X typically do better (M06 and ωB97X not yet in vibe-qc).
DFT-D3 in the loop¶
Adding dispersion for each functional (using
dispersion="d3bj"):
for functional in ("PBE", "BLYP", "B3LYP"):
r = run_job(
mol, basis="6-31g*",
method="rks", functional=functional,
dispersion="d3bj",
output=f"water_{functional}_d3",
write_molden_file=False,
)
print(f"{functional:8s} E_SCF = {r.energy:.6f} "
f"E_disp = {r.e_dispersion:+.6e} "
f"E_total = {r.energy_total:.6f}")
Dispersion is a small correction for isolated water (~0.7 mHa ≈ 0.4 kcal/mol of intra-molecular contribution) but dominates intermolecular binding in dimers, layered materials, and host-guest systems.
Sanity checks before you trust the number¶
Basis saturation. See basis-set convergence. A good functional on an inadequate basis is worse than a bad functional on a good one.
Grid quality. All numbers above use the default DFT grid (Treutler-Ahlrichs, ≈ 46k points/atom). For meta-GGAs and hybrids pushed to ~8 significant figures, bump
RKSOptions.grid.n_radialto 99 or 120.SCF tolerance. Defaults are tight (
conv_tol_energy = 1e-8 Ha). For convergence-sensitive properties (especially low-frequency vibrations) tightenconv_tol_gradtoo.
Theory — the functionals in detail¶
Tutorial 02 introduced the Kohn-Sham framework and Jacob’s ladder in the abstract. Here we look at the specific functionals used above, each of which represents a different philosophy for approximating \(E_\text{xc}[\rho]\).
LDA — the local-density approximation¶
LDA is the “at every point, pretend you’re at the matching point in a uniform electron gas” ansatz:
where \(\varepsilon_\text{xc}^\text{HEG}\) is the per-electron XC energy of
the homogeneous electron gas, known analytically for exchange
(Slater / Dirac formula) and numerically for correlation (Ceperley-
Alder quantum Monte Carlo, 1980; VWN parametrisation, 1980).
vibe-qc’s SVWN is Slater exchange + VWN5 correlation — the
canonical LDA. LDA over-binds bonds by ~0.2 Å of length and ~10
kcal/mol of dissociation energy on typical organics; now used mostly
for teaching, quick screens, and metallic solids.
GGA — adding the gradient¶
Generalised-gradient approximations (GGAs) lift the “uniform” assumption by adding \(|\nabla\rho|\) as a second argument:
PBE (Perdew-Burke-Ernzerhof 1996) is the non-empirical GGA — every parameter fixed by physical constraints (UEG limit, Lieb-Oxford bound, correct slowly-varying expansion). Workhorse for periodic calculations. BLYP (Becke 1988 exchange + Lee-Yang-Parr 1988 correlation) is an empirical GGA with different strengths — better for organic thermochemistry at the cost of transferability.
Hybrid functionals and self-interaction¶
Pure DFT has self-interaction error: the Coulomb term \(J[\rho]\) includes an electron’s interaction with itself, and the local exchange potential only partially cancels it. HF exchange cancels self-interaction exactly (the diagonal \(K_{ii} = J_{ii}\) term). Hybrid functionals mix in a fixed fraction \(\alpha\) of HF exchange:
B3LYP (Becke 1993, reparametrised Stephens et al. 1994) uses \(\alpha = 0.20\) with three empirical coefficients fit to a thermochemistry benchmark; the “3” refers to three parameters:
PBE0 (Adamo-Barone 1999) is parameter-free — PBE’s GGA with a quarter of HF exchange mixed in on rigorous adiabatic-connection grounds:
The tradeoff: hybrids are ~2× more expensive than pure DFT per SCF step (the HF-exchange sum scales \(\mathcal{O}(N^4)\)), but self- interaction is reduced and band gaps / barriers / HOMO levels are better. For organics where reaction energies matter, the cost is nearly always worth paying.
Why this is empirical, not systematic¶
Unlike coupled cluster (where CCSDTQ → CCSDT → CCSD(T) → CCSD → MP2 is a systematic hierarchy converging to exactness), there is no knob you can turn to make a DFT functional systematically more accurate. Different functionals are different physical models for the same unknown object. Benchmarking against CCSD(T) or experiment on a target system class is the only defensible way to choose. The GMTKN55 benchmark (Grimme’s group, 2017) is the canonical test set.
Resources¶
~250 MB peak RAM, ~5 s on one core (Apple M2 baseline) for the four-functional H₂O sweep. Hybrid functionals (B3LYP, PBE0, M06) add ~30% over a pure GGA at this size due to the HF exchange contribution to the Fock build; range-separated hybrids (\(\omega\)B97X-D) add another factor of ~2 because the long-range exchange goes through a separately-screened lattice sum.
References¶
LDA correlation parametrisation. S. H. Vosko, L. Wilk, and M. Nusair, “Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis,” Can. J. Phys. 58, 1200 (1980).
Uniform-gas correlation reference (QMC). D. M. Ceperley and B. J. Alder, “Ground state of the electron gas by a stochastic method,” Phys. Rev. Lett. 45, 566 (1980).
BLYP exchange + correlation. A. D. Becke, “Density-functional exchange-energy approximation with correct asymptotic behavior,” Phys. Rev. A 38, 3098 (1988); C. Lee, W. Yang, and R. G. Parr, “Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density,” Phys. Rev. B 37, 785 (1988).
PBE. J. P. Perdew, K. Burke, and M. Ernzerhof, “Generalized gradient approximation made simple,” Phys. Rev. Lett. 77, 3865 (1996); erratum ibid. 78, 1396 (1997).
Hybrid DFT, original. A. D. Becke, “A new mixing of Hartree- Fock and local density-functional theories,” J. Chem. Phys. 98, 1372 (1993); the three-parameter form (same author): “Density-functional thermochemistry. III. The role of exact exchange,” J. Chem. Phys. 98, 5648 (1993).
B3LYP as used in practice. P. J. Stephens, F. J. Devlin, C. F. Chabalowski, and M. J. Frisch, “Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields,” J. Phys. Chem. 98, 11623 (1994). This is the paper that defines “B3LYP” as every code now implements it (Becke’s 1993 half-and-half mixed VWN for correlation; Stephens et al. swap in LYP).
PBE0. C. Adamo and V. Barone, “Toward reliable density functional methods without adjustable parameters: the PBE0 model,” J. Chem. Phys. 110, 6158 (1999).
GMTKN55 benchmark set. L. Goerigk, A. Hansen, C. Bauer, S. Ehrlich, A. Najibi, and S. Grimme, “A look at the density functional theory zoo with the advanced GMTKN55 database for general main group thermochemistry, kinetics, and noncovalent interactions,” Phys. Chem. Chem. Phys. 19, 32184 (2017).
Next¶
Vibrational frequencies — how the functional affects IR spectra.
Dispersion corrections — the quick way to fix non-covalent interactions across any functional.